Technical Consulting & Program Management
The rigor of science,
the velocity of entrepreneurs.
We ship deliverables that speak for themselves:
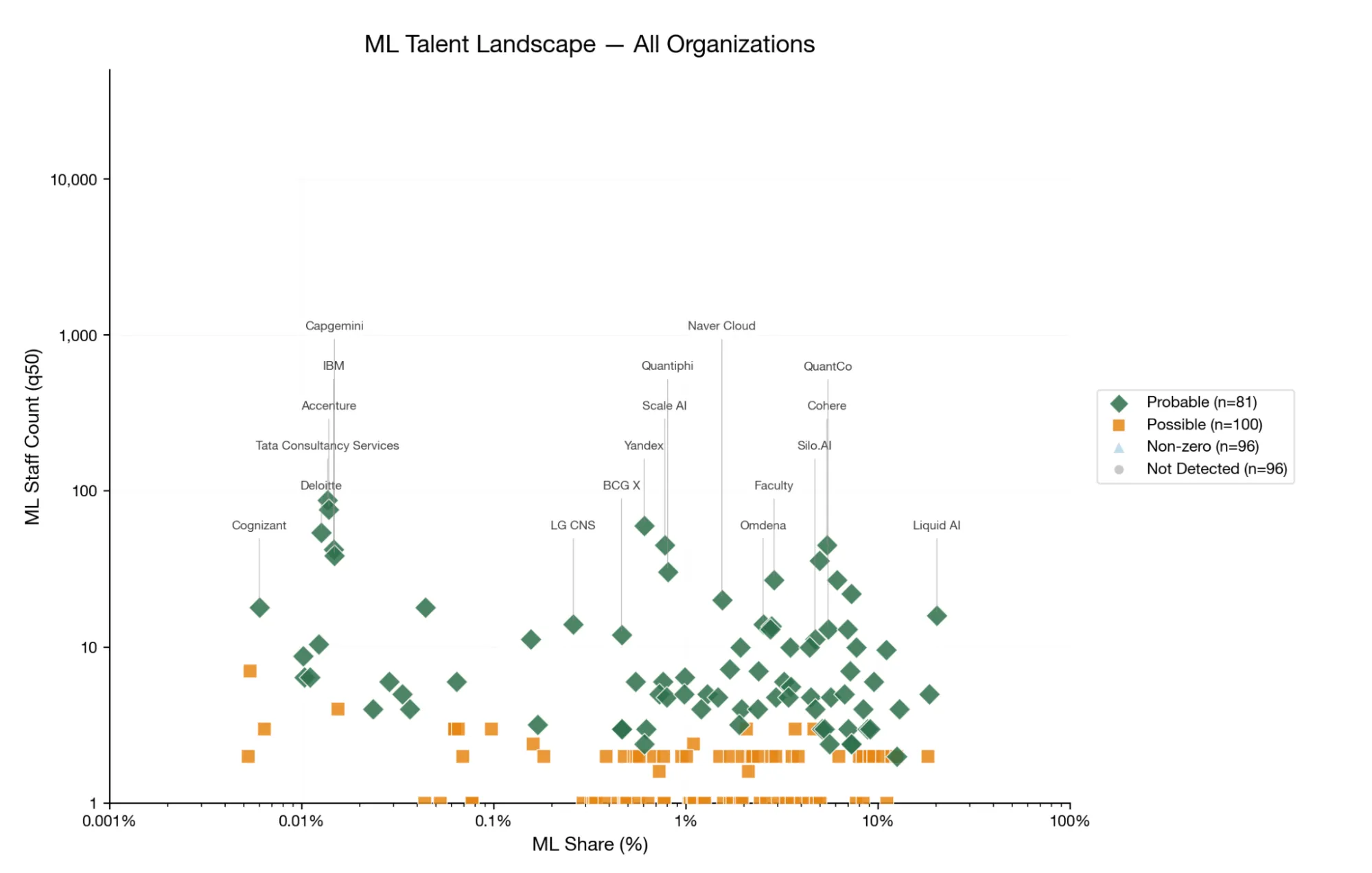
Nepenthe: ML Research Talent in IT Consultancies
Systematic estimate of technical ML research talent across 3 million IT consultancy employees, validated through work trials and AI agent benchmarks.

State of Brain Emulation Report 2025
A comprehensive assessment of 20 years of progress toward digitally replicating brains

The 2025 Peregrine Report
208 Expert Proposals for Reducing AI Risk
Featured Series
Drug Development Explainer
A concise introduction to drug development and its regulation. Get up to speed in ~1 hour on how pharma works—from the stakeholders and processes to the history that shaped today’s system.
Stakeholders
Regulators, sponsors, CROs, and the players who make it happen
02The Blueprint
From discovery through clinical trials to market approval
03Origins
How drug regulation evolved from tragedy to system
04Resources
Recommended literature and courses for deeper study
05Case Study: Alvea
The fastest biotech to reach human trials
06Conclusions
Key lessons and what the future might hold
Written in collaboration with James Smith and Kirsten Angeles.
More Writing
Recent Posts
On Meeting the Legitimate Needs of Mid-Career Talent
Investors and large foundations already meet the needs of senior talent — for themselves. Why mission-driven projects struggle to hire mid-career professionals, and what to fix.
Resurrecting MeCell Maps with Claude Code
One evening, 44 commits, and a decade-old project lives again
Comparing AI Labs and Pharmaceutical Companies
What the AI industry can learn from pharma: safety budgets, scientific rigor, and ecosystem checks and balances.
My human cell map illustration MeCell now in the public domain
My learning resource for students of the human cell available now for free in English, Spanish, Chinese and German.
The 100h clinical trial
A blueprint for 100-hour pharmaceutical response teams to deploy vaccines at outbreak sites. Speed is the decisive factor.
About

I'm Max.
I'm a physician who ended up building research infrastructure. During COVID I co-authored the proposal for Germany's $30M national pandemic study platform (NAPKON), then ran clinical operations as CMO at Alvea, a biotech startup that dosed its first patient 174 days after founding. In 2024 I started this company for technical consulting on responsible innovation at the intersection of biotech and AI. We assemble the right people and ship work that matters. Some of the output is available on this website.
Based in Germany, usually everywhere.